We’ve all heard of the “forgetful stoner”, and while some of us have better recall after excessive indulgence, others may have more difficulties. This is due to the modulation of certain signaling events within the neurons of cannabis users and over time differences in the structures of the brain responsible for memory formation. This article will explain what occurs at the structural and cellular level that corresponds with the hypomnesia associated with cannabis use.
Some Biological Concepts
Cannabinoid receptors are what are known as G-Protein Coupled Receptors (GPCRs). GPCRs are receptors that lead to signaling cascades, ultimately affecting the types of genes that are expressed in the cell. Once a ligand (a compound that has affinity for a receptor) binds to the receptor it can produce one of three responses. It can raise a receptor response above basal activity (agonist), cause a lack of response (antagonist), or cause activity to dip below basal levels (inverse agonist).
The binding of the ligand to the receptor leads to a signaling cascade that either decreases or increases the activity of a particular enzyme. These enzymes typically are responsible for the production of molecules known as secondary messengers. The amount of secondary messengers present in a cell dictates the activity of other enzymes called kinases and phosphatases. The role of kinases is to phosphorylate (add a chemical phosphate group) and the role of phosphatases is to dephosphorylate (remove a chemical phosphate group) onto downstream target proteins. These target proteins are usually transcription factors, proteins that bind DNA and promote the expression of certain genes within the cell. We can think of the chemical phosphate group added to these transcription factors as on and off switches for the expression of certain genes.
Ligand → Receptor → Ligand-Receptor Complex → Enzyme → Secondary Messenger Production → Kinase and Phosphatase Activity → Phosphorylation or Dephosphorylation of target proteins → Gene expression
The effect of THC on CB1 receptor signaling (simplified)
Now that there is a basic framework for our understanding of how G-Protein Coupled Receptors work, here’s how they apply to cannabinoid pharmacology. In this case we will consider the effect of the most well known constituent of cannabis, THC, as our ligand and its action at one of its most well known targets, the cannabinoid receptor CB1.
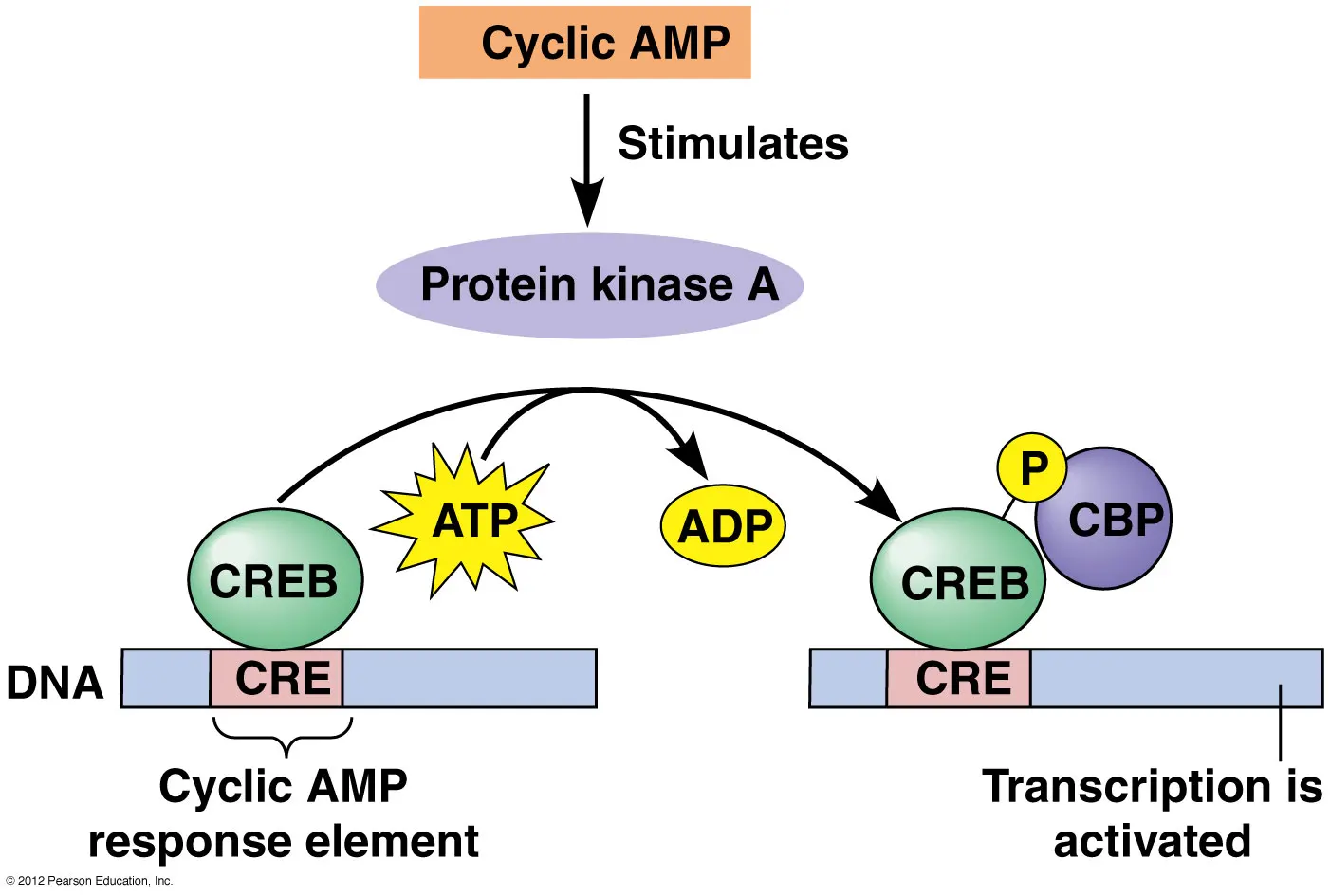
Once the cannabinoid receptor agonist THC binds to the CB1 receptor it triggers a signaling cascade as described above. The first step of this signaling cascade is a decrease in the function of an enzyme called adenylyl cyclase. This enzyme is responsible for the production our secondary messenger molecule which is called cyclic adenosine monophosphate (cAMP). The levels of cAMP regulate the activity of a kinase known as Protein Kinase A (PKA) and its associated phosphatase. This lowered activity of adenylyl cyclase causes lower levels of cAMP meaning that PKA activity is decreased while the activity of its associated phosphatase (nuclear protein phosphatase 2A) is increased. The target of interest for PKA and its associated phosphatase is called cAMP-response binding protein (CREB) [1]. CREB acts as a transcription factor binding to the DNA within the nucleus of a cell to promote the transcription of certain genes.
When CREB is phosphorylated it is in its “on-state” and will bind to certain regions of DNA promoting the expression of genes involved in memory. When CREB is not phosphorylated it is in its “off-state” and these memory genes are not actively being transcribed. It has been known for some time that CREB activity is required for the storage of memories, especially those associated with spatial and social learning [2-3]. Cannabinoids act on this signaling pathway by reducing cAMP levels as well as decreasing the amount of phosphorylated CREB (pCREB) in the hippocampus, resulting in a decrease in the expression of CREB-associated memory genes which is a partial cause of memory deficits in some cannabis users [4].
This is only a piece of the story, a more recent study by the Sackler Faculty of Medicine also using murine models says otherwise, finding elevated levels of pCREB. However, in their investigation they used ultra low doses of THC (.002 mg/kg), a dose significantly lower than most cannabis users would be receiving [5]. This difference in findings is likely due to the biphasic response of cannabinoids, meaning different doses can induce entirely different responses.
This is a ton of biological jargon to those who are non-science oriented so here is the breakdown of what goes on in the neurons of cannabis users:
THC binds to the CB1 receptor → ↓Adenylyl cyclase enzyme activity → ↓ cAMP → ↓PKA Activity and ↑Phosphatase Activity → ↓Phosphorylated CREB and ↑Non-Phosphorylated CREB → ↓ Expression of memory associated genes
Of course this is an extremely simplified overview of how this all works within the brain, so it would incorrect to assume that if an agonist of CB1 produces this response then an inverse agonist should produce the opposite. These signaling pathways are very complex and have many other components that I have left out for simplification.
There are other processes on the level of cell-to-cell communication that are modified. These alterations occur in a process called long-term depression (LTD) and occur specifically in the hippocampus, one of the structures responsible for memory formation [6].
If you’d like to learn more about how cannabinoids modulate hippocampal LTD I’ve also written a brief summary of their findings: http://sclabs.com/cannabinoids-and-memory/.
It has also been suggested that chronic cannabis use is associated with structural changes in the hippocampus, such as reduced gray matter and alterations in shape. One particular study showed that long-term cannabis users had a twelve percent reduction in hippocampal volume on average, however these findings may be flawed since there was a very small sample size used (n=15) [7-9]. If these changes are valid they could have an influence on memory formation in cannabis users. However a newer study refutes these findings, showing that daily cannabis use does not cause alterations in adolescents or adults. This study also used a much greater sample size than previous studies, comparing the brain morphology of twenty-nine adult users and non-users, as well as fifty adolescent users and non-users using high-resolution MRI scans [10].
Other aspects of cannabinoid signaling lead to memory deficits as well. One such aspect is the upregulation of COX-2 signaling which is an enzyme responsible for the conversion of arachidonic acid to prostaglandins and other prostanoids. This is why COX-2 inhibitors such as Tylenol have been shown to alleviate some of the memory deficits associated with cannabis use in mouse models [11].
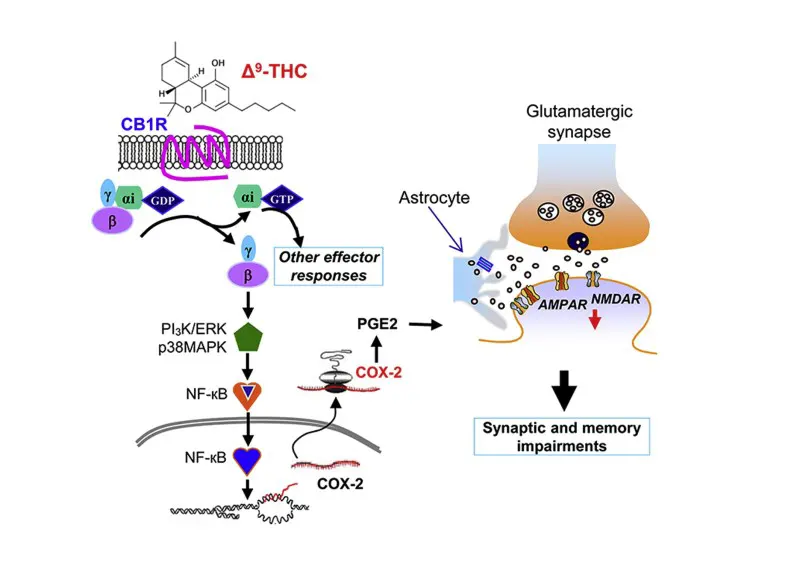
I have also written about these findings at the following link:
http://sclabs.com/preventing-cannabis-induced-memory-deficits-with-otc-pain-medication/
Another factor that contributes to memory deficits in cannabis users is its effects on REM sleep. It’s been known for some time that REM sleep is important for memory consolidation and cannabis use has been shown to decrease the time we spend in REM sleep [12-13]. It has also been speculated that when cannabis use is discontinued REM sleep rebounds and causing increased time in this phase of sleep. This could be an underlying reason as to why when cannabis users abruptly abstain from cannabis they dream more vividly.
Conclusion
There are many processes at hand that can lead to changes in memory in cannabis users. These changes occur at the molecular, cellular, and structural level of the hippocampus. There has been some work done to further our understanding of how these molecular deficits could be mitigated with compounds such as non-steroidal anti-inflammatory drugs (NSAIDs) as well as the use of cannabis with a higher CBD to THC ratio, however it is still unclear how THC induced changes in electrical activity could be prevented. As cannabis is becoming more widely accepted as a therapeutic option further research into tactics for the mitigation of memory deficits is certainly warranted.
References
- Wadzinski BE, Wheat WH, Jaspers S, et al. Nuclear protein phosphatase 2A dephosphorylates protein kinase A-phosphorylated CREB and regulates CREB transcriptional stimulation. Mol Cell Biol. 1993;13(5):2822-34
- Silva AJ, Kogan JH, Frankland PW, Kida S. CREB and memory. Annu Rev Neurosci. 1998;21:127-48.
- Kandel ER. The molecular biology of memory: cAMP, PKA, CRE, CREB-1, CREB-2, and CPEB. Mol Brain. 2012;5:14.
- Fan N, Yang H, Zhang J, Chen C. Reduced expression of glutamate receptors and phosphorylation of CREB are responsible for in vivo Delta9-THC exposure-impaired hippocampal synaptic plasticity. J Neurochem. 2010;112(3):691-702.
- Fishbein M, Gov S, Assaf F, Gafni M, Keren O, Sarne Y. Long-term behavioral and biochemical effects of an ultra-low dose of Δ9-tetrahydrocannabinol (THC): neuroprotection and ERK signaling. Exp Brain Res. 2012;221(4):437-48.
- Han J, Kesner P, Metna-laurent M, et al. Acute cannabinoids impair working memory through astroglial CB1 receptor modulation of hippocampal LTD. Cell. 2012;148(5):1039-50.
- Demirakca T, Sartorius A, Ende G, et al. Diminished gray matter in the hippocampus of cannabis users: possible protective effects of cannabidiol. Drug Alcohol Depend. 2011;114(2-3):242-5.
- Yücel M, Solowij N, Respondek C, et al. Regional brain abnormalities associated with long-term heavy cannabis use. Arch Gen Psychiatry. 2008;65(6):694-701.
- Solowij N, Walterfang M, Lubman DI, et al. Alteration to hippocampal shape in cannabis users with and without schizophrenia. Schizophr Res. 2013;143(1):179-84.
- Weiland BJ, Thayer RE, Depue BE, Sabbineni A, Bryan AD, Hutchison KE. Daily marijuana use is not associated with brain morphometric measures in adolescents or adults. J Neurosci. 2015;35(4):1505-12.
- Chen R, Zhang J, Fan N, et al. Δ9-THC-caused synaptic and memory impairments are mediated through COX-2 signaling. Cell. 2013;155(5):1154-65.
- Feinberg I, Jones R, Walker JM, Cavness C, March J. Effects of high dosage delta-9-tetrahydrocannabinol on sleep patterns in man. Clin Pharmacol Ther. 1975;17(4):458-66.
- Luo J, Phan TX, Yang Y, Garelick MG, Storm DR. Increases in cAMP, MAPK activity, and CREB phosphorylation during REM sleep: implications for REM sleep and memory consolidation. J Neurosci. 2013;33(15):6460-8.